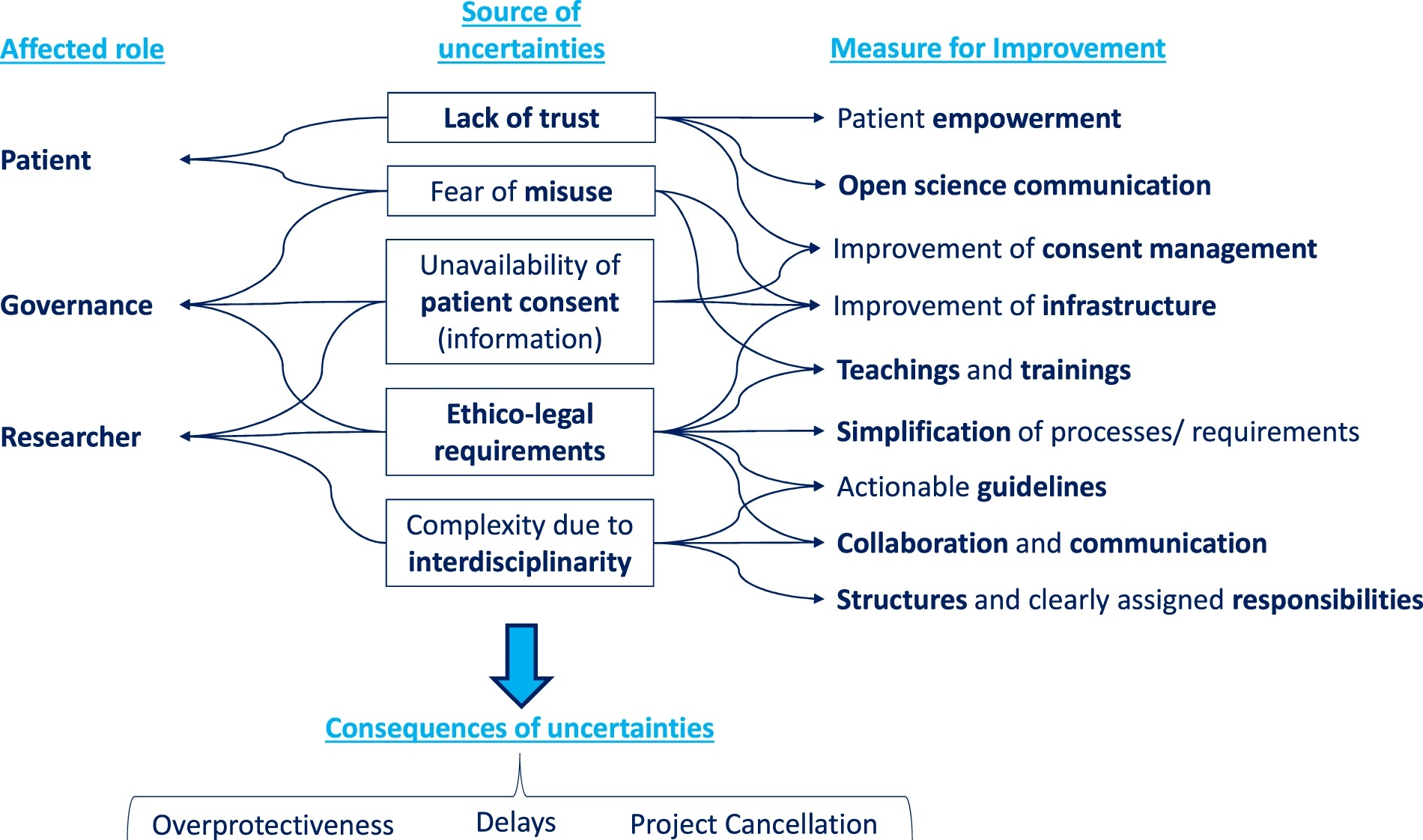

Leveraging the reuse of Real-World Data (RWD) in medical research comes with key objectives: i) advancing interoperability, ii) promoting harmonization, iii) ensuring data integrity, and iv) establishing protocols for data utilization and access. Yet, the complex legal framework around RWD hinders progress.
From a technical perspective, the absence of shared standards, user-friendly interfaces, structured data, and patient-centric clinical data systems poses challenges.
As highlighted in the insightful commentary paper "What prevents us from reusing medical real-world data in research", it is evident that a paradigm shift is needed in the way we collect and manage RWD. A critical first step in this direction is to implement data infrastructure that is compliant by design, both from a legal and technical perspective.
At AHDB, we recognize that addressing these challenges requires a fundamental cultural shift in the medical research landscape. This shift can be facilitated by an initial change in the technical paradigm currently used to handle medical RWD.
Follow our Linkedin page for more updates about our collaborations and initiatives